Graphene battery gives more power charging in less time
Vanadium Graphene battery sheet and ribbons
A lot of people when use mobile , tablet , laptop or any device depend in battery have main complaint battery discharge so fast and so slowly to recharge !
The reason behind that the most battery made from Lithium and lithium-ion battery (sometimes write Li-ion battery or LIB) have some problems
Li-Ion batteries have good capacity compared to their volume and weight if compared with another batteries materials and have a long life it have about 400–1200 cycles of charge and discharge, but There is a drawback, however: they take a long time to charge. Another drawback is their maximum power output. Li-Ion batteries may have a large capacity, but their maximum power output is limited because the risk of overheating.
Graphene Battery:
Graphene batteries are made by enhancing existing Li-Ion batteries, This is achieved by enriching the electrodes with graphene which changes their chemical and physical properties. The results are improved charge/discharge rate characteristics as well as improved capacity. Improved discharge rate means that graphene batteries have a higher maximum power output.
Graphene is not only being used for the electrodes of batteries, but for the active material itself. Graphene makes such a good battery material that the devices are called “supercapacitors”, i.e. batteries which hold enormous power and charge within a few seconds. Already two years ago graphene supercapacitors that store as much energy as nickel metal hydride (Ni: MH) batteries were demonstrated. in February this year, UCLA researchers caused shock waves when they showed a graphene mixture that can be coated onto the surface of a regular DVD. An ordinary DVD burner is then used to inscribe millions of super-capacitor circuits into the graphene layer. The layer can later be simply peeled off and transferred anywhere where a super powered battery is desired.
With such development, we might soon see ultra-thin flexible batteries which charge in less than a minute and could be integrated into clothes, paper, car dashboards or just about anywhere you want them.
Vanadium graphene to give more power:
Vanadium oxides offer high energy capacity when used in Li-Ion batteries. This is because they collect lithium ions like a sponge. The drawback of using vanadium oxide (vo5) or vanadium pentoxide (vo5) is that oxides in general are bad electrical conductors. The low conductivity results in a slower charge/discharge rate.
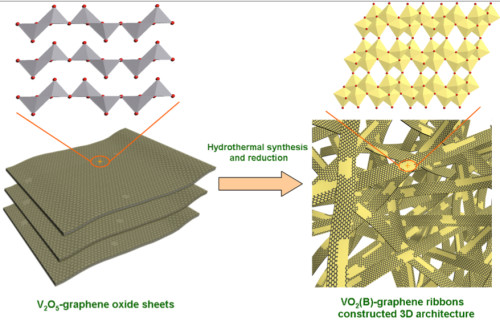
Scientists found a way to use graphene, which is an excellent conductor, as a structural backbone to which vanadium oxide is attached. This hybrid inherits good capacity properties from vanadium oxide and good conductivity from graphene which allows for a fast recharge.
Hybrid ribbons of vanadium oxide (vo5) and graphene may accelerate the development of high-power lithium-ion batteries suitable for electric cars and other demanding applications. The Rice University lab of materials scientist Pulickel Ajayan determined that the well-studied material is a superior cathode for batteries that could supply both high energy density and significant power density. The research appears online this month in the American Chemical Society journal Nano Letters.
The ribbons created at Rice are thousands of times thinner than a sheet of paper, yet have potential that far outweighs current materials for their ability to charge and discharge very quickly. Cathodes built into half-cells for testing at Rice fully charged and discharged in 20 seconds and retained more than 90 percent of their initial capacity after more than 1,000 cycles.
“This is the direction battery research is going, not only for something with high energy density but also high power density,” Ajayan said. “It’s somewhere between a battery and a supercapacitor.”

Graphene-coated ribbons of vanadium oxide, seen in a scanning electron microscope image
How vanadium graphene battery made?
The ribbons also have the advantage of using relatively abundant and cheap materials. “This is done through a very simple hydrothermal process, and I think it would be easily scalable to large quantities,” he said.
Ajayan said vanadium oxide has long been considered a material with great potential, and in fact, vanadium pentoxide has been used in lithium-ion batteries for its special structure and high capacity. But oxides are slow to charge and discharge, due to their low electrical conductivity. The high-conductivity graphene lattice that is literally baked in solves that problem nicely, he said, by serving as a speedy conduit for electrons and channels for ions.
The atom-thin graphene sheets bound to the crystals take up very little bulk. In the best samples made at Rice, fully 84 percent of the cathode’s weight was the lithium-slurping vo5, which held 204 milliamp hours of energy per gram. The researchers, led by Rice graduate student Yongji Gong and lead author Shubin Yang, said they believe that to be among the best overall performance ever seen for lithium-ion battery electrodes.
“One challenge to production was controlling the conditions for the co-synthesis of vo5 ribbons with graphene,” Yang said. The process involved suspending graphene oxide nanosheets with powdered vanadium pentoxide (layered vanadium oxide, with two atoms of vanadium and five of oxygen) in water and heating it in an autoclave for hours. The vanadium pentoxide was completely reduced to vo5, which crystallized into ribbons while the graphene oxide was reduced to graphene, Yang said. The ribbons, with a web-like coating of graphene, were only about 10 nanometers thick, up to 600 nanometers wide and tens of micrometers in length with a single-atom-thick coating of graphene. These ribbons are tens of micrometers long, and they have a very large specific surface area which allows for very fast diffusion of electrons and Li-Ions due to vo5’s ion-soaking properties and graphene’s high conductivity. These two properties combined allow for quick charge and discharge rates, as well as high maximum power outputs for these batteries.